According to the findings of a new study conducted by researchers at the United States Department of Veterans Affairs (VA), the antiviral medication known as Paxlovid, which reduces the risk of hospitalisation and mortality due to Covid-19, also reduces the risk of extended Covid.
A preprint of the study was made available online on Saturday. It consisted of an analysis of the electronic records of more than 56,000 veterans who were infected with Covid-19. Of them, more than 9,000 of them were treated with Paxlovid during the first five days of their illness.
According to the findings of the study, patients who were administered Paxlovid had a reduced risk of developing multiple long-term conditions by 26%. These conditions included heart disease, blood disorders, fatigue, liver disease, kidney disease, muscle pain, neurocognitive impairment, and shortness of breath.
This resulted in 2.3 fewer incidences of long-term Covid problems occurring per 100 patients three months following their diagnosis. Paxlovid also decreased the likelihood of requiring hospitalisation or passing away as a consequence of acute Covid-19.
According to the findings of the study, there was no statistically significant association between taking Paxlovid and the likelihood of developing long-term diseases such as cough or a new diabetes diagnosis.
The research paper was uploaded to the preprint server medRxiv; however, it has not yet been vetted by other researchers or published in a scholarly medical publication.
Patients who participated in the trial had a mean age of 65 and were given a diagnosis of Covid-19 between March 1 and June 30 in 2022. They all had at least one risk factor for developing severe Covid-19, such as older age, diabetes, or the fact that they were current smokers.
According to the study, Paxlovid was able to minimise the risk of lengthy Covid in people who had not been vaccinated, in those who had been vaccinated and boosted, and in people who were having either their first infection with Covid-19 or reinfection.
According to Dr Ziyad Al-Aly, chief of research and development at the VA St. Louis Health Care System and leader of the study, in a news release, “Paxlovid reduces the chance of severe COVID-19 in the acute phase, and now, we have evidence that it can also reduce the risk of protracted COVID.” This treatment has the potential to be a significant asset in the fight against the critical problem of extended COVID.
The study had several shortcomings, one of which is that the vast majority of the participants were white and male, which may reduce the usefulness of the findings to a broader audience. Although many people who have long Covid describe a wide variety of symptoms, the analysis only took into account the use of Paxlovid through the VA system. Also, the analysis only included a total of 12 long Covid disorders.
Since their first illness, millions of people who have had Covid-19 have struggled with a variety of persistent symptoms, but there is still no cure or treatment available for extended Covid.
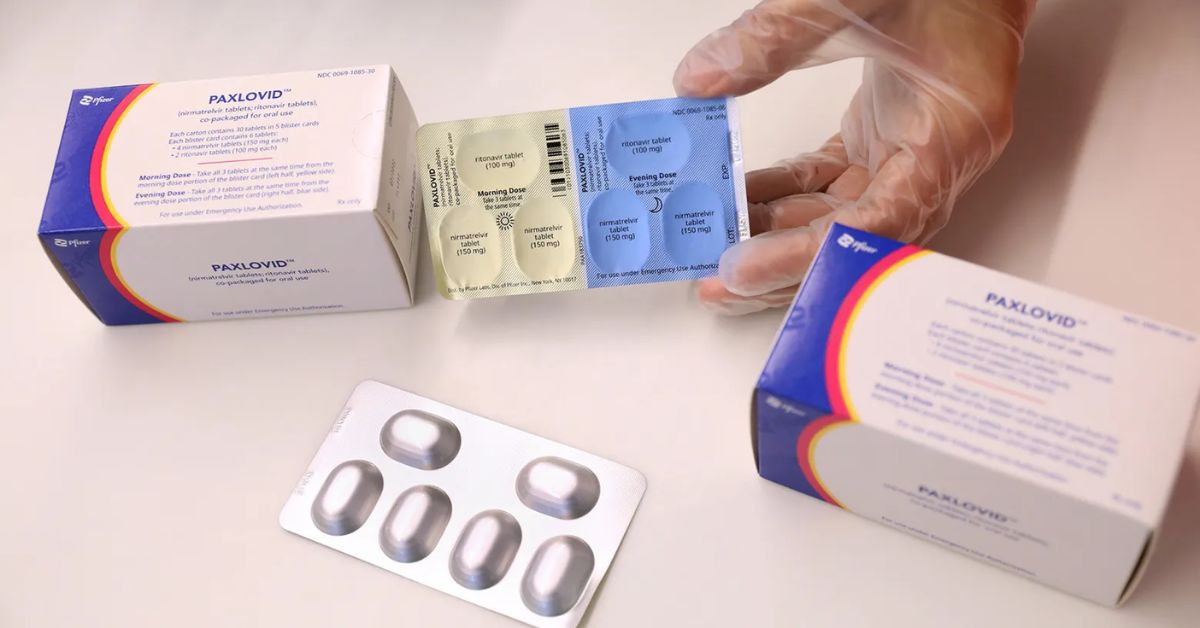
Paxlovid is an antiviral treatment for Covid-19 that combines nirmatrelvir, an antiviral that was developed more recently, with ritonavir, a medication that was developed more than a decade ago. It has been demonstrated to significantly lower the risk of hospitalisation and death in people at risk of severe COVID-19, and it is offered to people as young as 12 years old.
The medicine is manufactured by Pfizer and is administered orally over five days. It works best if started within five days of the onset of symptoms; researchers noted that it is not clear whether a longer duration or higher dose or both might reduce the risk of long Covid conditions even more, or if starting Paxlovid after acute illness caused by Covid-19 would reduce risk of long Covid.
Paxlovid works best when started within five days of the onset of symptoms. The National Institutes of Health announced a month ago that they would begin research on the drug Paxlovid as a potential treatment for individuals who are already experiencing long-term Covid.
According to what the authors of the VA study had written, “the totality of evidence suggests the need to improve uptake and utilisation of nirmatrelvir in the acute phase as a means of not only preventing progression to severe acute disease but also to reduce the risk of post-acute adverse health outcomes.”
If you enjoyed reading this post, please share your feedback with us in the comments box below. In addition, don’t forget to check back on our website Journalistpr.com frequently for the most recent updates. More Latest News: A Well-known Lawyer For Elon Musk Who Played A Key Role In Twitter’s Layoffs And Five States Have Measures About Abortion On Their Midterm Elections Ballots

Leave a Reply